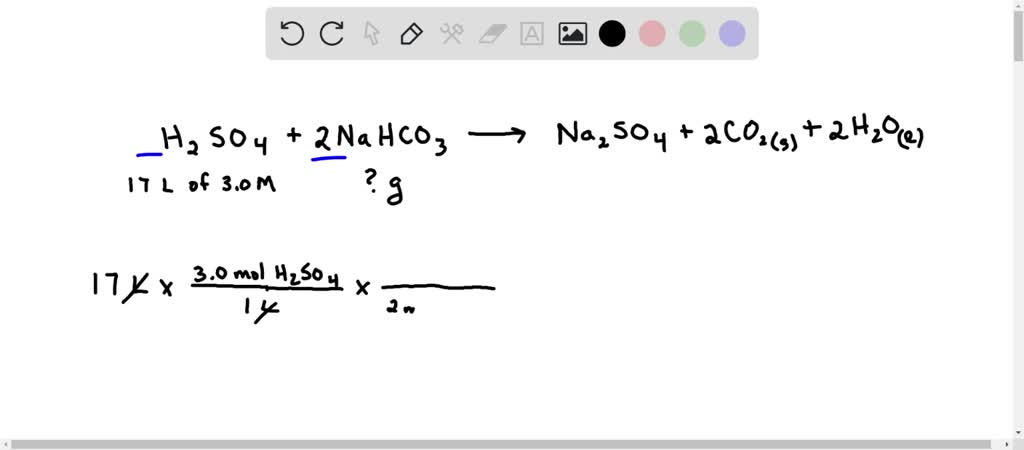
SOLVED:In a soda-acid fire extinguisher, concentrated sulfuric acid reacts with sodium hydrogen carbonate to produce carbon dioxide, sodium sulfate, and water. a. How many moles of sodium hydrogen carbonate would be needed
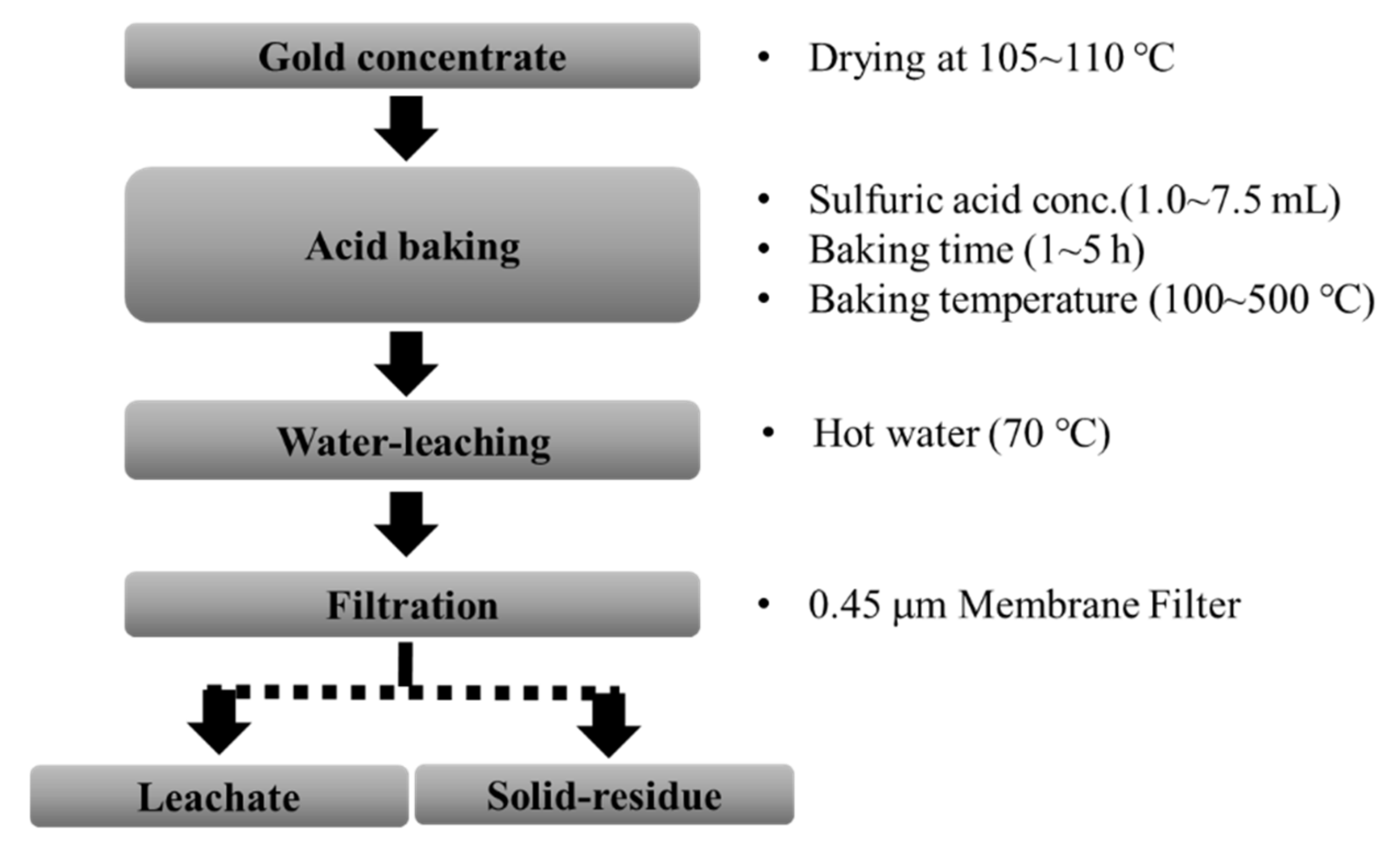
Minerals | Free Full-Text | Sulfuric Acid Baking—Water Leaching for Gold Enrichment and Arsenic Removal from Gold Concentrate | HTML
He literally just nutted in me and started shopping for fine pentaerythritol, 70% AR grade nitric acid,98% Sulfuric acid, acetone, filter paper, 300 ml beakers, a thermometer, distilled water, and baking soda. - )

Question Video: Determining the Concentration of Sulfuric Acid Via Titration with Sodium Carbonate | Nagwa

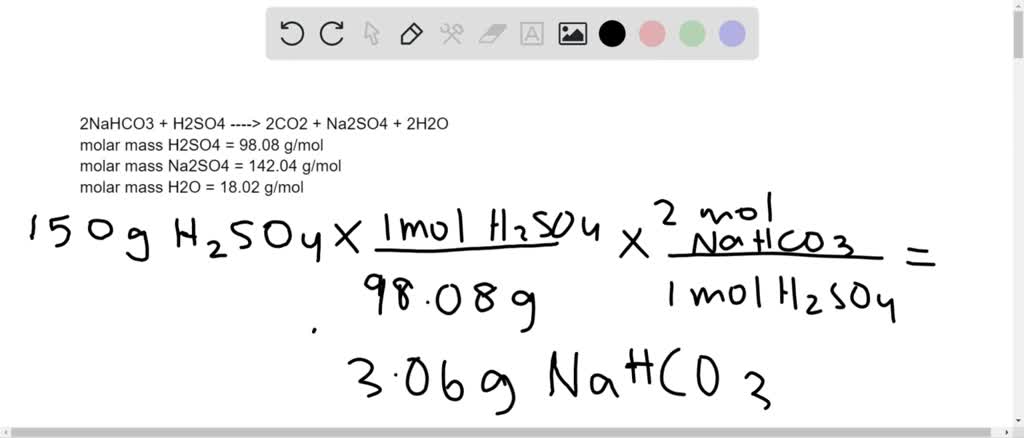
SOLVED: Aqueous solutions of sodium bicarbonate and sulfuric acid react to produce carbon dioxide according to the following equation: 2 NaHCOs(aq) + H,SO4(aq) 3 2CO2(g) + NazSO4(aq) + 2 HzO() If 105.4